What are the various regulatory review
Question: What are the various regulatory review pathways that the FDA can consider in order to release a vaccine (BLA, EUA, Expanded access)
Answer: The regulatory approval pathways are summarized below. The FDA will rely on the Vaccines and Related Biological Products Advisory Committee (VRBPAC) to make recommendations based on its evaluation of safety, efficacy, and appropriate use data available for the COVID-19 vaccine candidates.
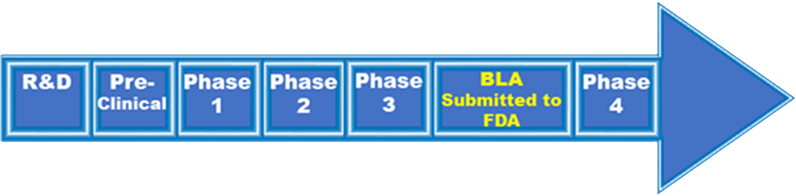
Biologics License Application (BLA):
Once preclinical and clinical development programs have been successfully completed, companies submit a BLA to the FDA. A BLA is a comprehensive submission which includes preclinical and clinical data and information, as well as details of the manufacturing process and facility(ies). The BLA is the official request for permission to introduce a biologic product, including a vaccine, into interstate commerce.
Emergency Use Authorization (EUA):
- EUAs helped speed access to COVID-19 diagnostic tests, N95 respirators, and remdesivir.
- COVID-19 vaccines: FDA prefers phase-3 studies be completed. EUA sooner could impair efficacy + safety determination.
Expanded access:
This allows investigational drugs, biologics, or medical devices that have not yet been approved or cleared by FDA to be used for a patient with an immediately life-threatening condition or serious disease for treatment outside of clinical trials when no comparable or satisfactory alternative therapy options are available.
Currently, expanded access is one pathway for use of COVID-19 convalescent plasma for patients with serious or immediately life-threatening COVID-19 disease who are not eligible for or who are unable to participate in randomized clinical trials.
Additional Resources:
- Understanding the Regulatory Terminology of Potential Preventions and Treatments for COVID-19
- FDA BLA Resources
- FDA EUA Guidance: https://www.fda.gov/media/97321/download
- FDA Vaccines and Related Biological Products Advisory Committee https://www.fda.gov/advisory-committees/blood-vaccines-and-other-biologics/vaccines-and-related-biological-products-advisory-committee
